胰蛋白酶-EDTA溶液(0.25%:0.02%)
产品简介:
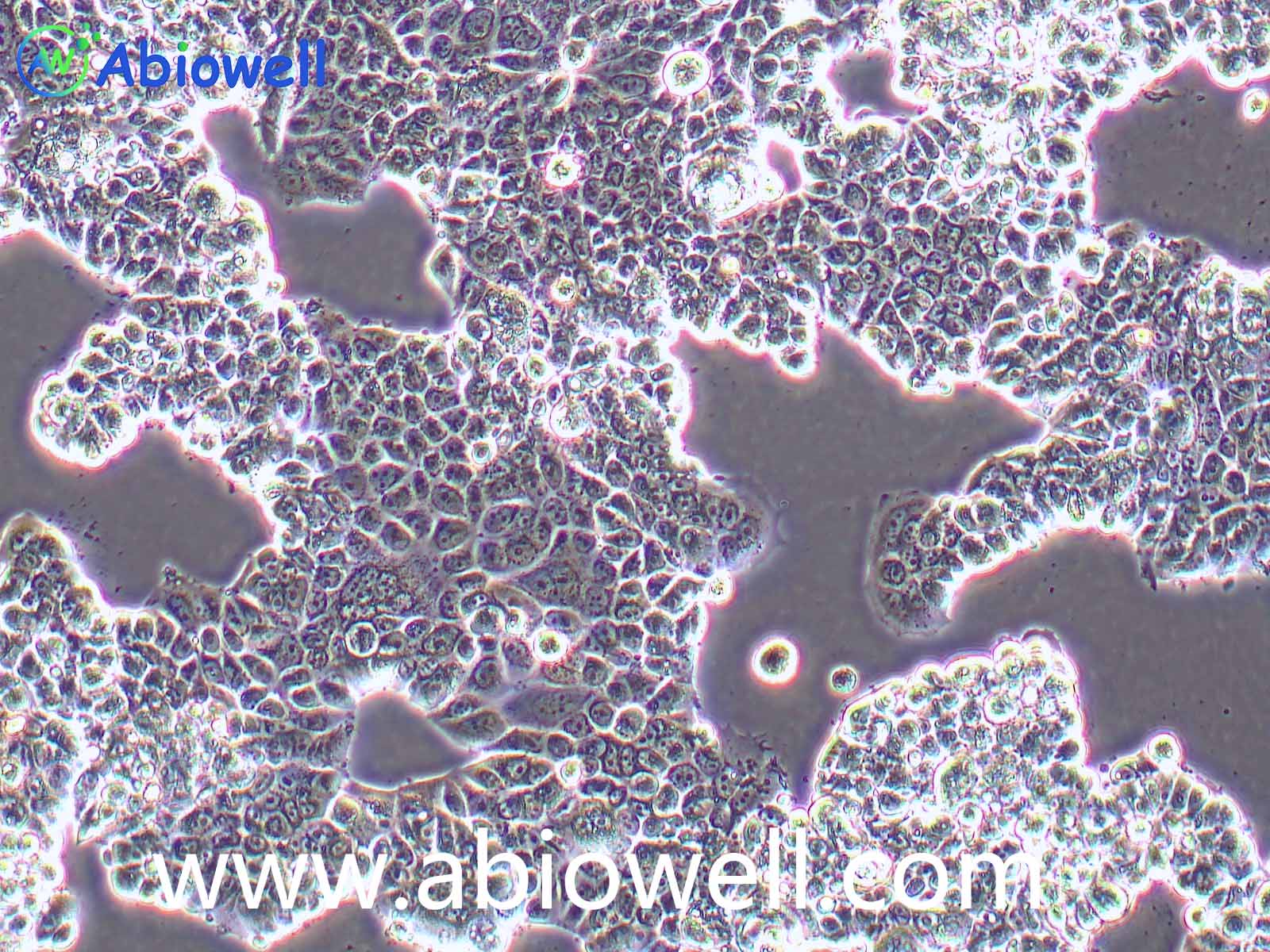
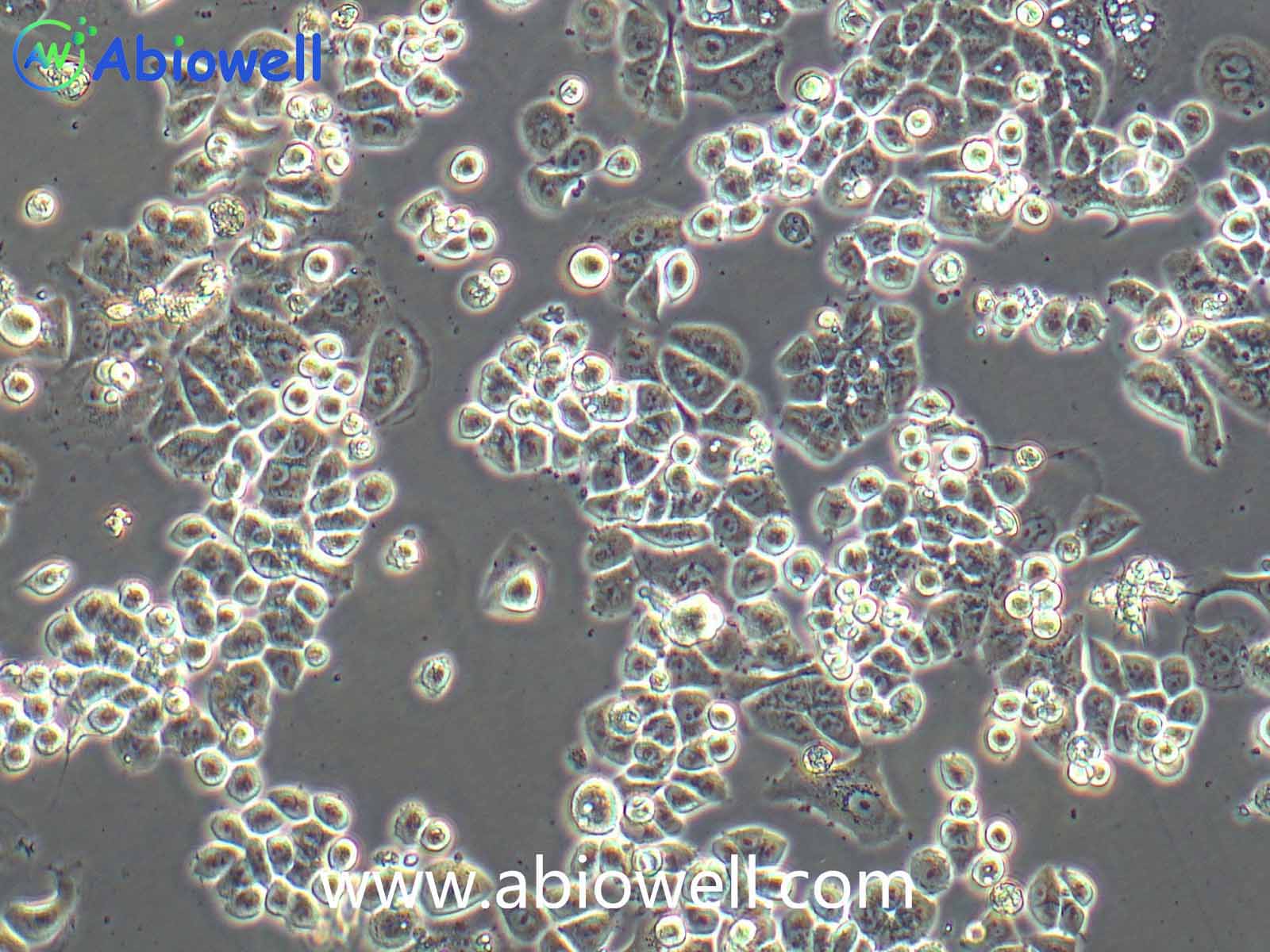
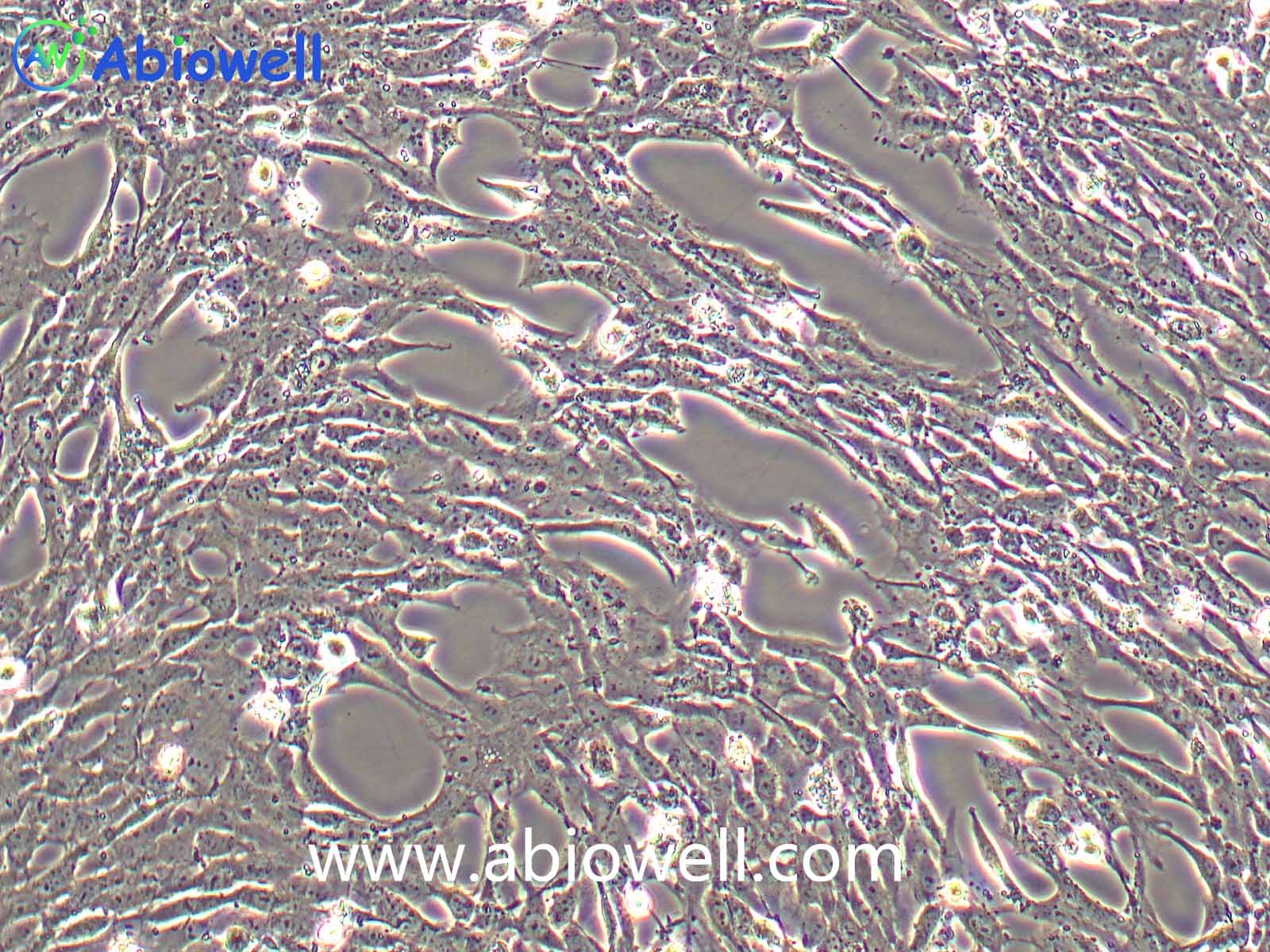
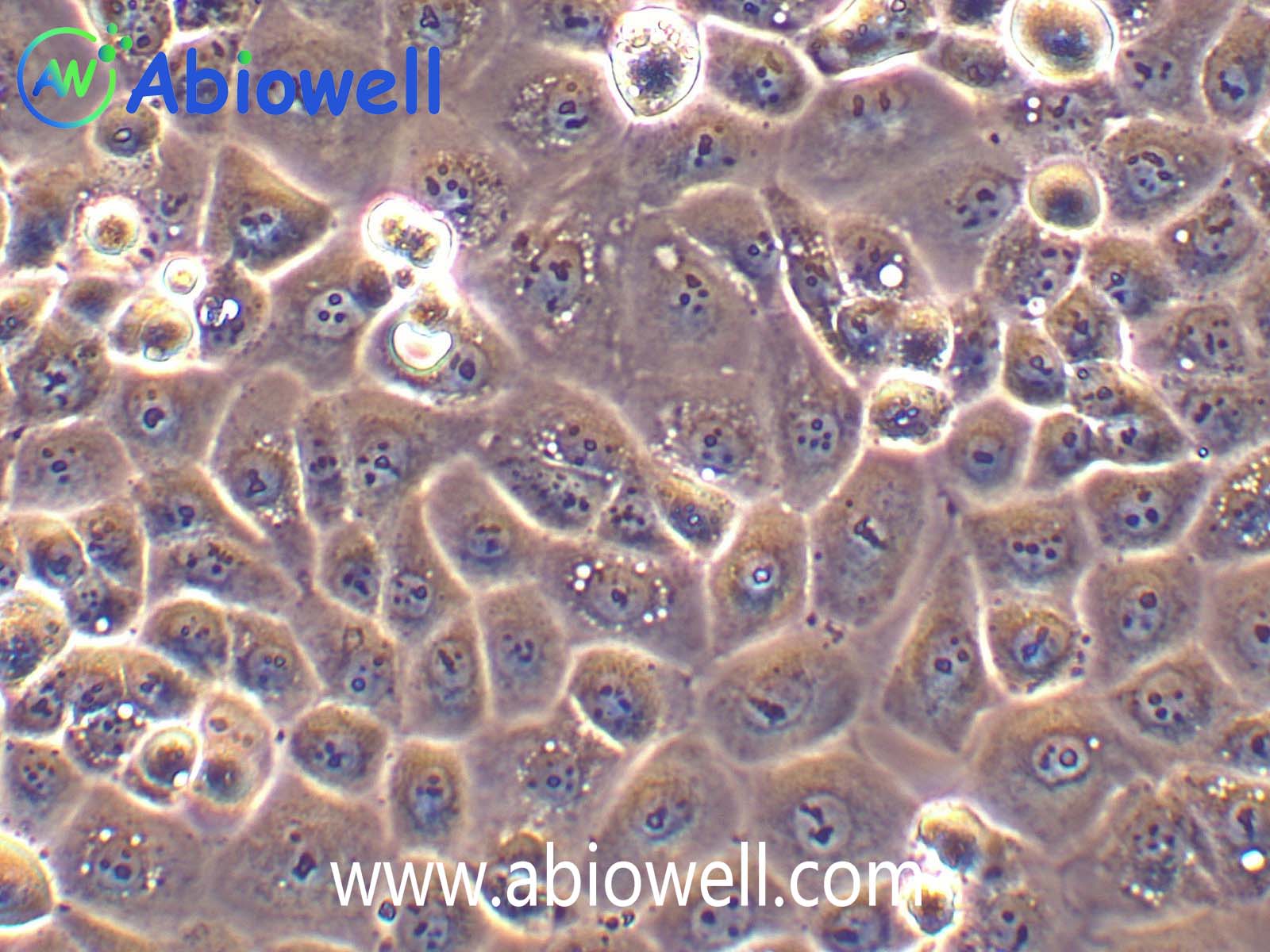
胰蛋白酶(Trypsin)是由胰脏产生没有活性的胰蛋白酶原分泌到小肠后,小肠内的肠肽酶会活化该酶原,形成胰蛋白酶。胰蛋白酶的特点在于已经活化的胰蛋白酶,能够继续活化更多胰蛋白酶原,这种过程即自动催化。胰蛋白酶在小肠工作,它会将蛋白质水解为肽,进而分解为氨基酸,其最适温度约为37℃。
Trypsin-EDTA Solution(0.25%:0.02%)由0.25%胰酶、0.02%EDTA等组成,经过滤除菌。本试剂可以直接用于培养细胞的消化,或者一些组织的消化,通常室温下1min左右就可以消化下大多数贴壁细胞。
自备材料:
1、 PBS、Hanks液或无血清培养液
2、 显微镜
3、 离心机
操作步骤(仅供参考):
1、 贴壁细胞的消化
① 吸除培养液,用无菌PBS、Hanks液或无血清培养液洗涤细胞1次,以去除残余的血清。
② 加入少量Trypsin-EDTA Solution,略盖过细胞即可,室温放置0.5~2min,不同的细胞消化时间有所不同。
③ 显微镜下观察,细胞明显收缩,并且肉眼观察培养皿底部发现细胞的形态发生明显的变化;或者用枪吹打细胞发现细胞刚好可以被吹打下来,吸除胰酶细胞消化液。加入含血清的完全细胞培养液,吹打下细胞,即可直接用于后续实验。
④ 如果发现消化不足,则加入Trypsin-EDTA Solution重新消化。
⑤ 如果发现细胞消化时间过长,未及吹打细胞,细胞已经有部分直接从培养器皿底部脱落,直接用胰酶细胞养液把细胞全部吹打下来。1000~2000g离心1min,沉淀细胞,尽量去除胰酶细胞消化液后,加入含血清的完全培养液重新悬浮细胞,即可用于后续实验。
2、 组织的消化
① 不同的组织需要消化的时间相差很大,通常以消化后可以充分打散组织为宜。
注意事项:
1、 尽量减少反复冻融的次数,以免失效。
2、 在使用Trypsin-EDTA Solution过程中,要特别注意避免消化液被细菌污染。
3、 Trypsin-EDTA Solution消化细胞时间不宜过长,否则细胞铺板后生长状况会较差。
4、 为了您的安全和健康,请穿好实验服并佩戴一次性手套和口罩操作。
5、 本产品仅限于专业人员的科学研究用,不得用于临床诊断或治疗,不得用于食品或药品,不得存放于普通住宅内。